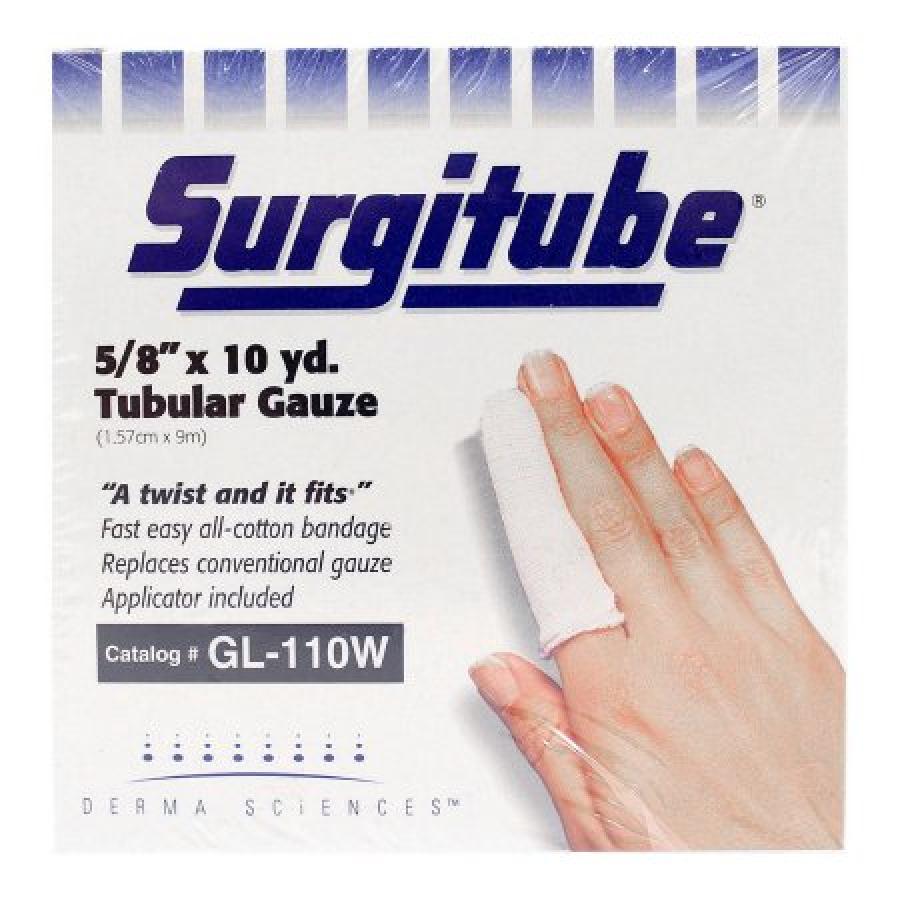
Integra LifeSciences SurgiTubes Tubular Retainer Dressings, Non-Sterile, White, Cotton, Size 1, 5/8 Inch x 10 Yard, 1 Count, #GL110W
Integra LifeSciences SurgiTubes Tubular Retainer Dressings, Non-Sterile, White, Cotton, Size 1, 5/8 Inch x 10 Yard, 1 Count, #GL110W
$6.80
Estimated Delivery Wednesday, April 8th.
- Product Details
- Features
- Q&A
Integra LifeSciences Corporation Surgitube Tubular Bandage is a premium-grade bandage renowned for its optimal wound care and support capabilities. Its open mesh design fosters airflow, promoting wound healing and preventing skin maceration. Non-raveling material allows for customizable cutting without unraveling, ensuring a secure fit. Applicator-free application simplifies dressing changes. Soft, absorbent cotton provides comfort and breathability. The bandage's versatility extends to various wound types, including burns, lacerations, abrasions, and post-operative sites. Its dimensions (5/8 inch x 10 yards) provide ample coverage, while its white color facilitates monitoring wound exudate and dressing changes. The Integra LifeSciences Corporation Surgitube Tubular Bandage empowers healthcare professionals and individuals with an effective and convenient wound management solution, promoting healing, preventing infections, providing support, and minimizing discomfort.
UOM : EA
UPC : 00817583023261
Application : Tubular Retainer Dressing
Color : White
COO_1 : Unknown
Dimensions : 5/8 Inch X 10 Yard
Material : Cotton
Size : Size 1
Sterility : NonSterile
Target Area : Small Finger / Toe
Type : Tubular
UNSPSC Code : 42311500
GTIN: 416926255804
MPN: GL110W
SKU: M313741
Item ID: 427405




















 Phone:
Phone:  6am to 5pm Monday to Friday Pacific Time
6am to 5pm Monday to Friday Pacific Time